Recently, the Drug Administration (Ministry of Health) has received letters from T&G Law Firm (authorized by Bayer Intellecture Property GmbH) reporting on the detection of product samples suspected of being counterfeit drugs. . These products have the following information on the label:
first. Stivarga 40mg (film kapli tablet, Regorafenib – Treatment drug) cancer rectal, cancer liver)
2. Xarelto 10mg (film kapli tablet, Rivaroksaban – Prevention of thromboembolism, stroke)
3. Xarelto 15mg (film kapli tablet, Rivaroksaban)
4. Xarelto 20mg (film kapli tablet, Rivaroksaban)
Samples of products suspected to be counterfeit drugs discovered by T&G Law Firm are sold on the market and some websites (https://healthyungthu.com, https://nhathuoclp.com, https://thuocdactrigan.com). According to the Company’s report and compared with the drug samples provided by T&G Law Firm, the above-mentioned products have different signs compared to the corresponding samples of Stivarga 40mg, Xarelto 10mg/15mg/20mg produced by Bayer. Manufacturing AG, Bayer Vietnam Co., Ltd. or Zuellig Pharma Vietnam Co., Ltd.’s Branches import and distribute.
To ensure the safety of users, the Drug Administration of Vietnam recommends the Department of Health of the provinces and centrally-run cities, the Health Departments and Bayer AG:
1. Notify business establishments and drug-using establishments about the characteristics and distinguishing signs between suspected counterfeit products and drugs manufactured by Bayer AG, by Bayer Vietnam Co., Ltd. or its branches. Zuellig Pharma Vietnam Co., Ltd. imports as follows:
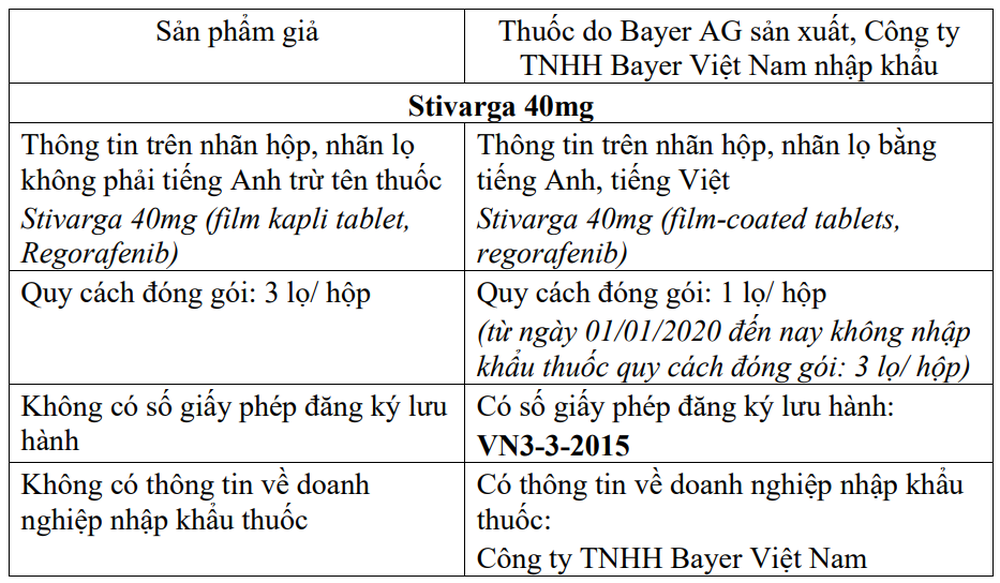
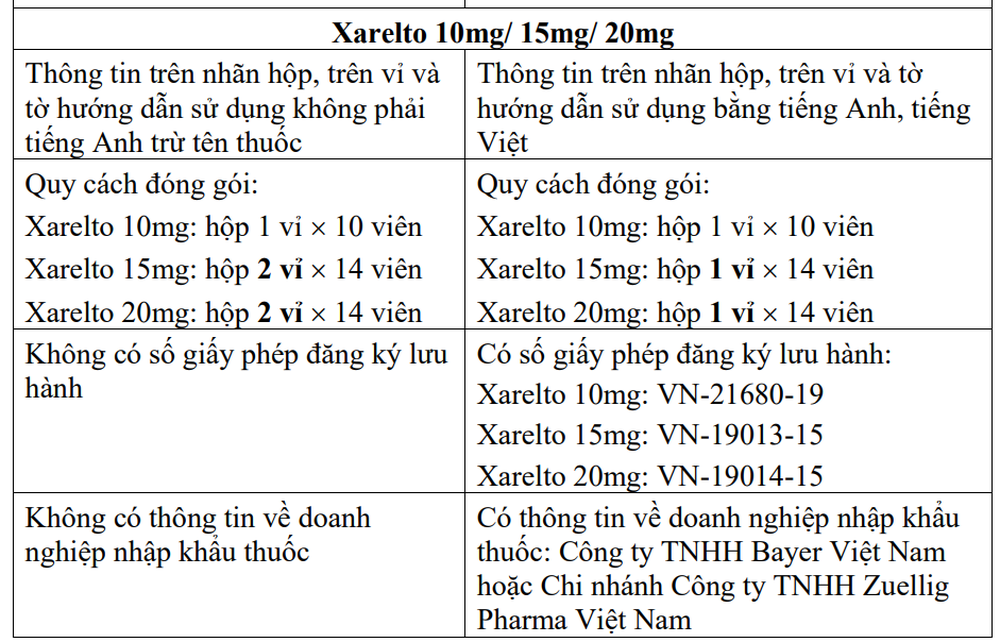
2. Coordinating with media agencies to inform drug dealers and users and the public to know so as not to trade or use counterfeit products on the label of Stivarga 40mg (film kapli tablet, Regorafenib). ), Xarelto 10mg (film kapli tablet, Rivaroksaban), Xarelto 15mg (film kapli tablet, Rivaroksaban)/ Xarelto 20mg (film kapli tablet, Rivaroksaban) and the above identification signs.
3. Request the manufacturer of Bayer AG or the drug registration facility of Stivarga 40mg, Xarelto 10mg, Xarelto 15mg, Xarelto 20mg to provide relevant documents and documents and promptly report it directly to the Drug Administration of Vietnam according to regulations. requested in Official Dispatch No. 1524/QLD-CL dated March 7, 2022 of the Drug Administration of Vietnam.
at Blogtuan.info – Source: Soha.vn – Read the original article here



